Peer Reviewed Clinical Journal Publishes Lexaria Bioscience's Human CBD Study Results
KELOWNA, BRITISH COLUMBIA / ACCESSWIRE / September 17, 2019 / Lexaria Bioscience Corp. (OTCQX:LXRP)(CSE:LXX) (the “Company” or “Lexaria”), a global innovator in drug delivery platforms, is pleased to announce that final study results of the 2018 human clinical study evaluating CBD delivery and effectiveness using its patented DehydraTECHTM powered TurboCBD™ capsules have been published in the peer reviewed medical journal, “Advances in Therapy”. Advances in Therapy focuses on clinical medicine and pharmaceutical research and has been published continually since 1984.
Key findings highlighted in the publication were as follows:
- CBD in-plasma levels with the TurboCBD™ 90 mg dose were significantly higher than with the generic 90 mg dose at both 90 and 120 min (p < 0.05);
- Only the TurboCBD™ 90 mg dose was elevated (i.e., greater than placebo) at 30 min (p < 0.05) and remained elevated (i.e., greater than placebo) at 4 h (p < 0.01);
- Mean arterial blood pressure (MAP) in a subgroup analysis was significantly decreased with only the TurboCBD™ 90 mg dose relative to baseline (p < 0.05); and
- Cerebral perfusion analysed by an index of conductance in the middle cerebral artery (MCA) in this subgroup was significantly higher with only the TurboCBD™ 90 mg dose relative to baseline (p < 0.001).
The study was conducted and well tolerated in 12 healthy young male athletes and the investigators concluded that further studies are warranted, noting in particular that “further acute and chronic dosing investigations in older and cerebrovascular-compromised patients may shed light on the vascular and clinical impacts of increased CBD bioavailability.”
Lexaria’s recent receipt of two granted patents for “treating certain conditions including, but not limited to heart disease, neurological diseases such as Alzheimer’s, Parkinson’s, schizophrenia” and others is timely considering the demonstrated effectiveness of DehydraTECH technology in increasing cerebral blood flow, and thus perhaps an increased effectiveness at delivering certain drugs to the brain.
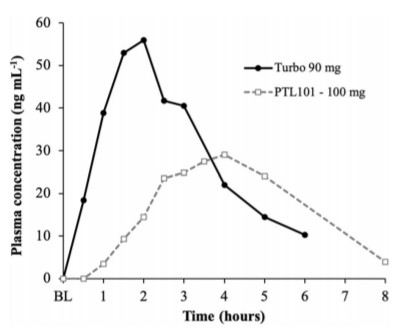
Finally, it is worth noting that the publication also references a separate recent study published by other investigators who evaluated a comparable CBD dose with bioabsorption findings representative of the cannabinoid edibles industry sector in general. The differences in blood concentration between the TurboCBD 90 mg dose and the competitor’s 100 mg dose are shown in the graph above, where time to maximum plasma CBD concentration with the competitive formulation required ~4 hours whereas it was evidenced with the TurboCBD™ 90 mg dose within 90-120 min. The rate of bioabsorption of the CBD with the competitive 100 mg formulation was virtually nondetectable within 30 minutes of administration.
The peer-reviewed study is available at https://rdcu.be/bQZZi and https://www.ncbi.nlm.nih.gov/pubmed/31512143
About Lexaria
Lexaria Bioscience Corp. is a global innovator in drug delivery platforms. Its patented DehydraTECH™ drug delivery technology changes the way Active Pharmaceutical Ingredients enter the bloodstream, promoting healthier ingestion methods, lower overall dosing and higher effectiveness for lipophilic active molecules. DehydraTECH increases bio-absorption; reduces time of onset; and masks unwanted tastes for orally administered bioactive molecules including cannabinoids, vitamins, non-steroidal anti-inflammatory drugs (NSAIDs), nicotine and other molecules. Lexaria has licensed DehydraTECH to multiple companies in the cannabis industry for use in cannabinoid beverages, edibles and oral products; and to a world-leading tobacco producer for the development of smokeless, oral-based nicotine products. Lexaria operates a licensed in-house research laboratory and holds a robust intellectual property portfolio with 16 patents granted and over 60 patents pending worldwide.
 For regular updates, connect with Lexaria on Twitter (https://twitter.com/lexariacorp)
For regular updates, connect with Lexaria on Twitter (https://twitter.com/lexariacorp)
 and on Facebook https://www.facebook.com/lexariabioscience/
and on Facebook https://www.facebook.com/lexariabioscience/
FOR FURTHER INFORMATION PLEASE CONTACT:
Lexaria Bioscience Corp.
Alex Blanchard, Communications Manager
(250) 765-6424 ext. 202
Or
NetworkNewsWire (NNW)
www.NetworkNewsWire.com
FORWARD-LOOKING STATEMENTS
This release includes forward-looking statements. Statements which are not historical facts are forward-looking statements. The Company makes forward-looking public statements concerning its expected future financial position, results of operations, cash flows, financing plans, business strategy, products and services, competitive positions, growth opportunities, plans and objectives of management for future operations, including statements that include words such as "anticipate," "if," "believe," "plan," "estimate," "expect," "intend," "may," "could," "should," "will," and other similar expressions are forward-looking statements, including but not limited to: that any additional patent protection will be realized or that patent achievements will deliver material results. Such forward-looking statements are estimates reflecting the Company's best judgment based upon current information and involve a number of risks and uncertainties, and there can be no assurance that other factors will not affect the accuracy of such forward-looking statements. Factors which could cause actual results to differ materially from those estimated by the Company include, but are not limited to, government regulation and regulatory approvals, managing and maintaining growth, the effect of adverse publicity, litigation, competition, scientific discovery, the patent application and approval process and other factors which may be identified from time to time in the Company's public announcements and filings. There is no assurance that existing capital is sufficient for the Company's needs or that it will be able to raise additional capital. There is no assurance the Company will be capable of developing, marketing, licensing, or selling edible products containing cannabinoids, nicotine or any other active ingredient. There is no assurance that any planned corporate activity, scientific research or study, business venture, letter of intent, technology licensing pursuit, patent application or allowance, consumer study, or any initiative will be pursued, or if pursued, will be successful. There is no assurance that any of Lexaria’s postulated uses, benefits, or advantages for the patented and patent-pending technology will in fact be realized in any manner or in any part. No statement herein has been evaluated by the Food and Drug Administration (FDA). Lexaria-associated products are not intended to diagnose, treat, cure or prevent any disease.
The CSE has not reviewed and does not accept responsibility for the adequacy or accuracy of this release.
SOURCE: Lexaria Bioscience Corp.
View source version on accesswire.com:
https://www.accesswire.com/559820/Peer-Reviewed-Clinical-Journal-Publishes-Lexaria-Biosciences-Human-CBD-Study-Results
Released September 17, 2019
